2025 International Myeloma Society Annual Meeting – Toronto, Canada | Virtual
First Author: Yair Levy
Session Information: 9/17/2025-9/20/2025
Poster Transcript
Introduction:
- Teclistamab was the first-in-class B-cell maturation antigen (BCMA) x CD3 bispecific antibody approved for the treatment of relapsed/refractory multiple myeloma (RRMM) that demonstrated an overall response rate (ORR) of 63%, after a median follow-up of 30.4 months in the MajesTEC-1 trial1
- Most U.S. patients with multiple myeloma (MM) are treated in community oncology clinics, where practice patterns may differ from academic centers2
- However, real-world data on teclistamab has been largely reported from academic centers with step-up dosing (SUD) conducted primarily in inpatient settings3-6
Objective
- To describe the clinical characteristics, SUD patterns, safety, and effectiveness of teclistamab in patients with RRMM treated at Texas Oncology, a large U.S. community oncology practice network
Methods:
Study design and data source
- This retrospective, observational study used patient charts and electronic medical record data for adult patients with RRMM treated with teclistamab at Texas Oncology’s practices
Study population
- Adult patients with ≥1 record of MM diagnosis based on the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) code (C90.0x) and ≥1 record for teclistamab between 10/26/2022 and 12/31/2024 were included
- Patients who received teclistamab in a clinical trial were excluded
- The date of the first observed diagnosis was considered the initial MM diagnosis date, and the date of Teclistamab initiation was defined as the index date
- Eligible patients were followed from the index date to the last activity, death, or data cutoff (12/31/2024)
- Tec-1 eligible subgroup: patients eligible for teclistamab per US Prescribing Information (USPI), defined as meeting the following criteria:
- Prior triple class exposed (TCE), including a proteasome inhibitor, an immunomodulatory drug, and an anti-CD38 monoclonal antibody
- Four prior lines of therapy prior to teclistamab initiation
- Eastern Cooperative Oncology Group (ECOG) performance status of 0-1
- No exposure to anti-BCMA therapy (ciltacabtagene autoleucel, idecabtagene vicleucel, elranatamab, or belantamab mafodotin) prior to teclistamab initiation
Data analysis
- All variables were summarized descriptively for all patients (the overall cohort) and for the Tec-1 eligible subgroup
- Time-to-event outcomes were estimated using the Kaplan-Meier method
Results:
Demographics and clinical characteristics
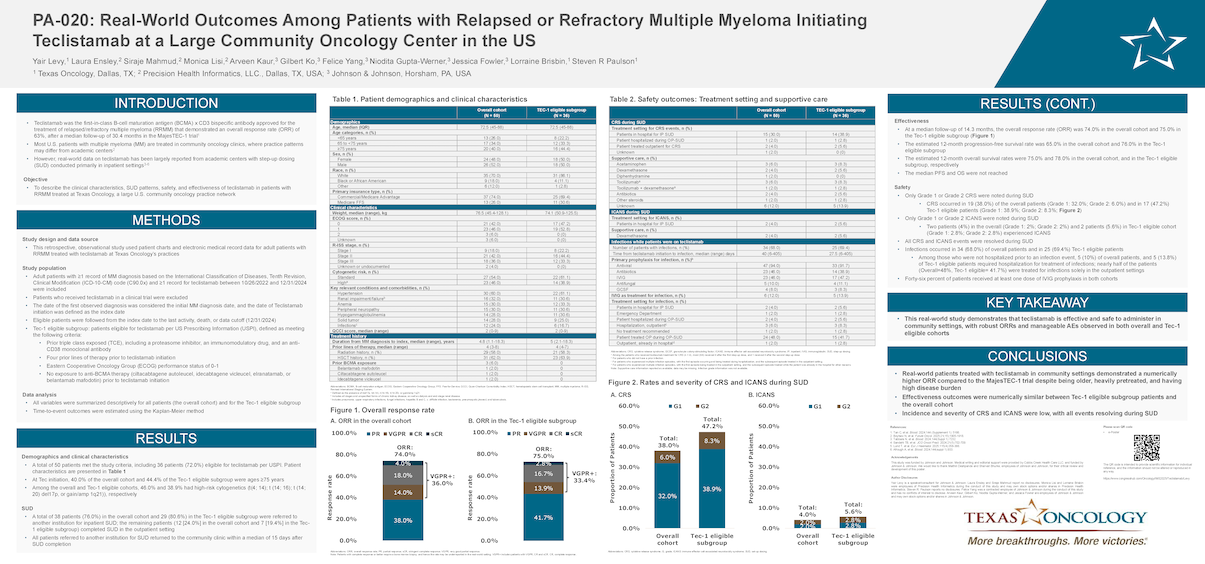
- A total of 50 patients met the study criteria, including 36 patients (72.0%) eligible for teclistamab per USPI. Patient characteristics are presented in Table 1
- At Tec initiation, 40.0% of the overall cohort and 44.4% of the Tec-1 eligible subgroup were ages ≥75 years
- Among the overall and Tec-1 eligible cohorts, 46.0% and 38.9% had high-risk cytogenetics (t(4; 14); t (14; 16); t (14; 20) del17p, or gain/amp 1q21)), respectively
SUD
- A total of 38 patients (76.0%) in the overall cohort and 29 (80.6%) in the Tec-1 eligible subgroup were referred to another institution for inpatient SUD; the remaining patients (12 [24.0%] in the overall cohort and 7 [19.4%] in the Tec-1 eligible subgroup) completed SUD in the outpatient setting
- All patients referred to another institution for SUD returned to the community clinic within a median of 15 days after SUD completion